Spravato Therapy
FDA-APPROVED relief for when oral anti-depressants aren’t enough
What Is SPRAVATO®?
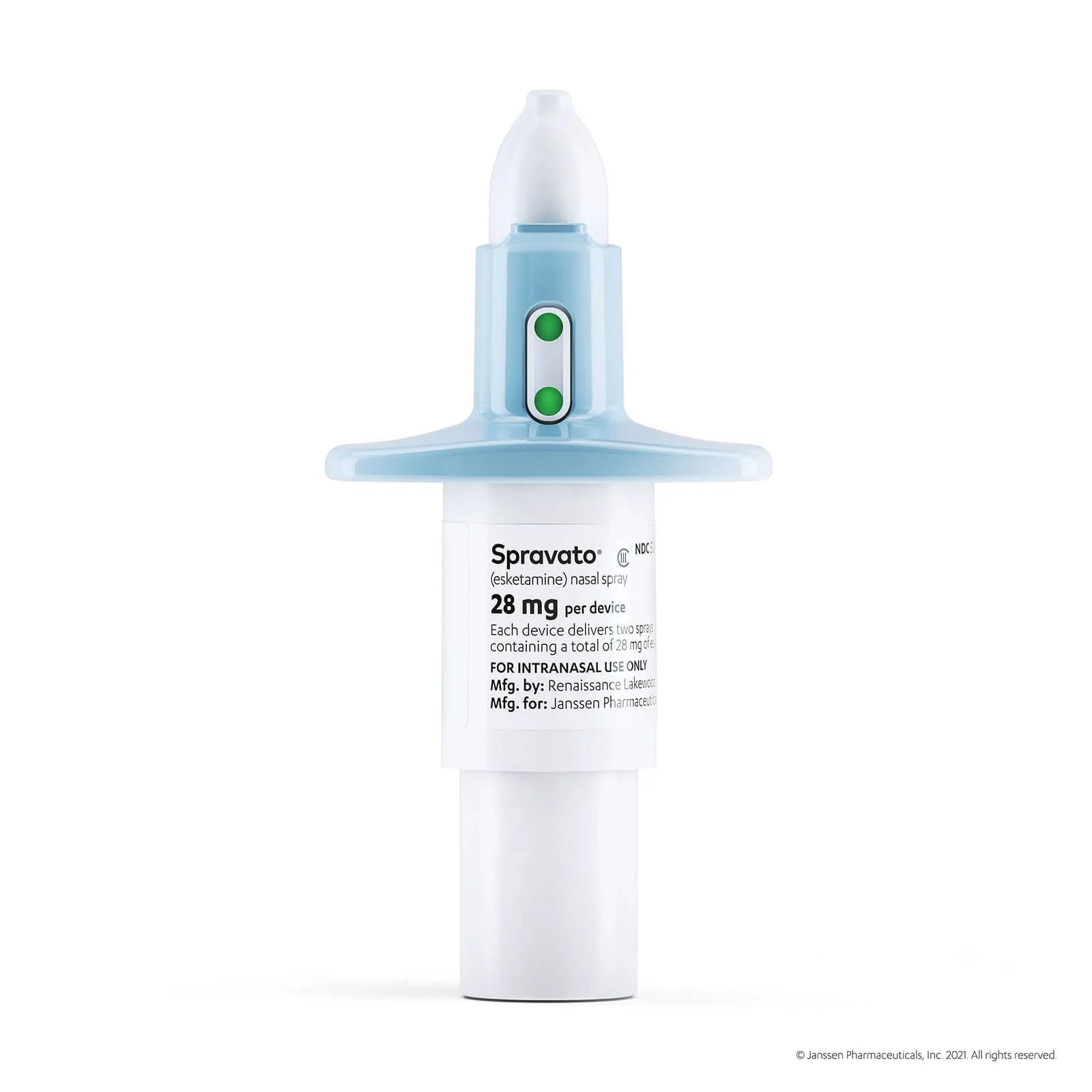
Spravato is a prescription medication (CIII Nasal Spray), for use in conjunction with an oral antidepressant. Spravato( esketamine), is an FDA approved medication used to treat Treatment- resistant depression.
SPRAVATO® nasal spray works differently than other medications to target depressive symptoms. SPRAVATO® targets the N-methyl-D-aspartate (NMDA) receptor. The exact way that SPRAVATO® works is unknown
Spravato may cause sleepiness (sedation), fainting, dizziness, spinning sensation, anxiety, or feeling disconnected from yourself, thoughts, feelings, space, and time (dissociation).
Who should take Spravato?
Adults with treatment-resistant depression (TRD)
Adults with major depressive disorder (MDD) with suicidal thoughts or actions.
Who should NOT take Spravato?
Anyone trying to relieve or prevent pain (anesthetic).
A replacement for hospitalization if your healthcare provider determines that hospitalization is needed.
Children
Contemporary oral antidepressants are widely understood to alleviate depression by elevating neurotransmitter levels in specific brain regions that influence mood. In contrast, Spravato focuses on the N-Methyl-D-Aspartate (NMDA) receptor, representing a departure from the mechanisms of action seen in currently prescribed oral antidepressants.
Esketamine, being the enantiomer of ketamine and an NMDA antagonist, operates by augmenting levels of the neurotransmitter glutamate and a growth factor known as BNDF.
Side Effects and Symptoms
Risk Evaluation and Mitigation Strategy (REMS):
Because of the risks for sedation, dissociation, and abuse and misuse, Spravato is only available through the restricted program called REMS. Spravato can only be administered at healthcare settings certified in the Spravato Rems program. It is used to closely monitor patients and maintain safety protocol.
Treatment Plan
The treatment plan typically involves an induction phase, where patients receive the nasal spray twice a week for 4 weeks, and a maintenance phase, where the frequency may be reduced.
Spravato is not a pain reliever or an aesthetic.
Side effects include dissociation, dizziness, nausea, sleepiness, feeling anxious, lack of energy, feeling very happy or excited. See the Spravato medication guide for complete safety information.
Getting Started: What to Expect?
-
1. Come in and relax in our chair

-
2. The spray is administered in your nasal cavity

-
3. Relax while our team supervises

-
4. Make sure to have someone drive you home!

FAQs
How long does a session last?
1
Approximately two hours, including administration and monitoring.
Can I stay on my current medications?
2
Yes. SPRAVATO® is taken in combination with your oral antidepressant (for MDD with suicidal thoughts) or alone (for TRD).
Is this an outpatient treatment?
3
Yes. SPRAVATO® is taken in combination with your oral antidepressant (for MDD with suicidal thoughts) or alone (for TRD).
Who may not be eligible?
4
Those with uncontrolled hypertension, history of aneurysms or strokes, certain sensitivities, or those who are pregnant or breastfeeding. We’ll help you assess safety during your evaluation.

Get Started Now on Your Journey of Healing
We are here to help. Our team has decades of combined experience in compassionate medical and mental health. Get in touch with us today to talk about building your plan.
